今年年初,新冠肺炎疫情在世界范围内快速暴发。新冠肺炎感染性强,感染后的重型、危重型患者病死率高,尚无特效治疗方法。而细胞治疗作为一种新型治疗技术,在新冠肺炎的临床研究治疗中发挥了重要作用。
近日,《新型冠状病毒肺炎细胞治疗的关键策略和研究进展》在最新一期《中华细胞与干细胞杂志(电子版)》上刊登发表。文章从新型冠状病毒发病机理和细胞治疗的选择策略、MSC治疗冠状病毒的潜在机制及其治疗同类疾病的临床研究进展、针对COVID-19的细胞治疗临床研究现状以及细胞治疗在COVID-19临床研究中的难点几个方面,总结并分析了细胞治疗新型冠状病毒肺炎最新研究进展。
该文章由湖北省华中科技大学同济医学院附属协和医院血液科、国家干细胞工程产品产业化基地、中国人民解放军总医院第五医学中心感染病医学部、国家感染性疾病临床医学研究中心联合撰写。
论文原文
一、新型冠状病毒发病机理和细胞治疗的选择策略
如何根据COVID-19的临床指征特别是CSS和ARDS选择合适的细胞对COVID-19进行干预和治疗,可以从现有诊疗指南中得到一些启发[2]。诊疗指南中对于适用药物的探索开发主要基于如下两点:一方面可以"老药新用",如已获批上市且其在人体内的代谢特征、使用剂量、潜在疗效和副作用都明确的抗病毒药物。同理,可以从过往细胞治疗CSS、ARDS等有关疾病的临床治疗研究中筛选可行的细胞种类;另一方面,追本溯源,明确治疗机理,进行精确打击,例如洛匹那韦/利托那韦(lopinavir/ritonavir)和目前备受期待的瑞德西韦(remdesivir)等。不同细胞在病毒免疫中的角色大不相同,病毒侵入人体后,单核细胞、NK细胞是第一道防线,随后巨噬细胞吞噬病毒传递抗原,T细胞和B细胞被抗原分泌的细胞因子激活,对抗并清除感染细胞,使人体恢复正常。目前,国内外在细胞治疗冠状病毒肺炎领域主要聚焦在异体间充质干细胞(mesenchymal stem cell,MSC)和自然杀伤(nature killer,NK)细胞上。有文献报道,体外和动物实验均已验证,NK细胞对SARS、H5N1、埃博拉、寨卡、登革热和HIV等多种突发病毒感染疾病有很好的抑制和清除能力[18,19,20]。但还未见在临床试验中直接使用异体NK细胞治疗上述病毒性传染病,其主要风险在于无法确切控制可能产生的免疫排异反应和免疫过激反应。但脐带血来源的NK细胞由于细胞来源广泛,并且免疫原性较低,可能是潜在的主要候选之一[21]。(图1)
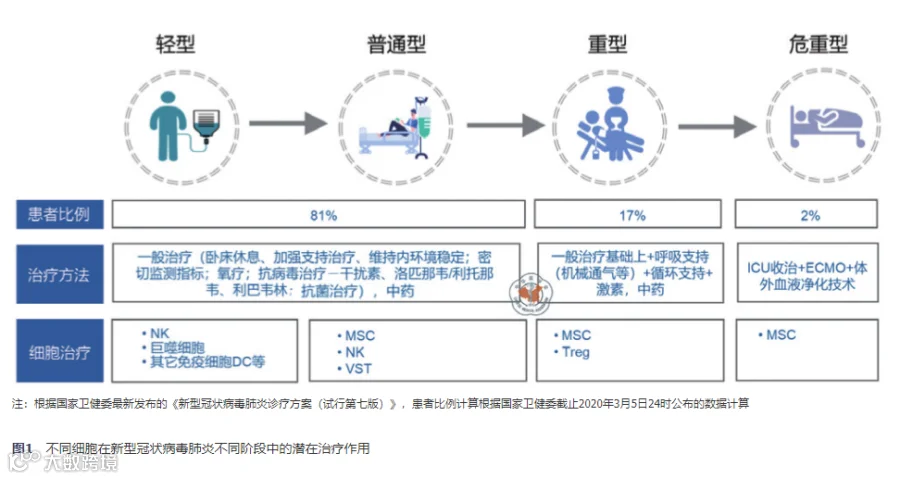 图1 不同细胞在新型冠状病毒肺炎不同阶段中的潜在治疗作用
图1 不同细胞在新型冠状病毒肺炎不同阶段中的潜在治疗作用
二、MSC治疗冠状病毒的潜在机制及其治疗同类疾病的临床研究进展
(一)免疫功能正常化作用(normalization)
(二)支持修复作用
三、针对COVID-19的细胞治疗临床研究现状

表1 Clinicaltrials和ChiCTR注册的干细胞临床研究
四、细胞治疗在COVID-19临床研究中的难点
五、总结与展望
志谢 本文在此感谢张宇博士提供了重要的帮助和支持,并共同完成了本文的发表工作
参考文献
1
国家卫生健康委员会. 国家卫生健康委关于新型冠状病毒肺炎暂命名事宜的通知[EB/OL].(2020-02-07)[2020-06-23]. http://www.gov.cn/zhengce/zhengceku/2020-02/08/content_5476248.htm.
2
国家卫生健康委员会.新型冠状病毒感染的肺炎诊疗方案(试行第七版) [EB/OL].(2020-03-03)[2020-06-23].http://www.nhc.gov.cn/yzygj/s7653p/202003/46c9294a7dfe4cef80dc7f5912eb1989.shtml.
3
国家卫生健康委员会.中华人民共和国国家卫生健康委员会公告[EB/OL].(2020-01-20)[2020-06-23].http://www.nhc.gov.cn/jkj/s7916/202001/44a3b8245e8049d2837a4f27529cd386.shtml.
4
世界卫生组织WHO. 关于2019新型冠状病毒疫情的《国际卫生条例(2005)》突发事件委员会第二次会议的声明[EB/OL].(2020-01-30). [2020-06-23].https://www.who.int/zh/news-room/detail/30-01-2020-statement-on-the-second-meeting-of-the-international-health-regulations-(2005)-emergency-committee-regarding-the-outbreak-of-novel-coronavirus-(2019-ncov).
5
SohrabiC, AlsafiZ, O'NeillN, et al. World Health Organization declares global emergency: a review of the 2019 novel coronavirus (COVID-19)[J]. Int J Surg, 2020, 76:71-76.
6
ChenW, PeterWH, FrederickGH, et al. A novel coronavirus outbreak of global health concern[J].Lancet, 2020, 395(10223):470-473.
7
ZhuN, ZhangD, WangW, et al. A novel coronavirus from patients with pneumonia in China, 2019[J]. N Engl J Med, 2020, 382(8):727-733.
8
VincentJM, KoopmansM, DoremalenNV, et al. A novel coronavirus emerging in China-key questions for impact assessment[J]. N Engl J Med, 2020, 382(8):692-694.
9
XuZ, LeiS, WangYJ, et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome[J]. Lancet Respir Med, 2020, 8(4):420-422.
10
LeeKY. Pneumonia, acute respiratory distress syndrome, and early immune-modulator therapy[J].Int J Mol Sci, 2017, 18(2):388.
11
ChenNS, ZhouM, DongX, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in wuhan, China:a descriptive study[J]. Lancet, 2020, 395(10223):507-513.
12
BauerTT, EwigS, RodloffAC, et al. Acute respiratory distress syndrome and pneumonia: a comprehensive review of clinical data[J]. Clin Infect Dis, 2006, 43(6):748-756.
13
HuangC, WangY, LiX, et al. Clinical features of patients infected with 2019 novel coronavirus in wuhan, China[J]. Lancet, 2020, 395(10223):497-506.
14
WangD, HuB, HuC, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in wuhan, China[J]. JAMA, 2020, 323(11):1061-1069.
15
NeelapuSS, TummalaS, KebriaeiP, et al. Chimeric antigen receptor T-cell therapy-assessment and management of toxicities[J]. Nat Rev Clin Oncol, 2018, 15(1):47-62.
16
XuX, HanM, LiT, et al. Effective treatment of severe COVID-19 patients with tocilizumab[J]. Proc Natl Acad Sci U S A, 2020, 117(20): 10970-10975.
17
A study to evaluate the safety and efficacy of tocilizumab in patients with severe COVID-19 pneumonia(COVACTA) (EB/OL) (2020-03-25) [2020-06-23].https://clinicaltrials.gov/ct2/show/study/NCT04320615.
18
WilliamGG, SubbaraoK, BrianM, et al. Mechanisms of host defense following severe acute respiratory Syndrome-Coronavirus (SARS-CoV) pulmonary infection of mice[J]. J Immunol, 2004,173(6):4030-4039.
19
JansanCA, de GeusED, van HaarlemDA, et al. Differential lung NK cell responses in avian influenza virus infected chickens correlate with pathogenicity[J]. Sci Rep, 2013, 3(1):2478.
20
ParsonsMS, BouletS, SongR, et al. Mind the gap: lack of association between KIR3DL1*004/HLA-Bw4-induced natural killer cell function and protection from HIV infection[J]. J Infect Dis, 2010,202Suppl 3: S356-360.
21
SarvariaA, JawdatD, MadrigalJA, et al. Umbilical cord blood natural killer cells, their characteristics, and potential clinical applications[J]. Front Immunol, 2017, 8:329.
22
DominiciM, Le BlancK, MuellerI, et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement[J]. Cytotherapy, 2006,8(4):315-317.
23
KramperaM, GalipeauJ, ShiY, et al. Immunological characterization of multipotent mesenchymal stromal cells--The international society for cellular therapy(ISCT)working proposal[J]. Cytotherapy,2013, 15(9):1054-1061.
24
LoyH, KuokDIT, HuiKPY, et al. Therapeutic implications of human umbilical cord mesenchymal stromal cells in attenuating influenza a(H5N1) virus-associated acute lung injury[J]. J Infect Dis,2019, 219(2):186-196.
25
LeeJW, FangX, KrasnodembskayaA, et al. Concise review: Mesenchymal stem cells for acute lung injury: role of paracrine soluble factors[J]. Stem Cells, 2011, 29(6):913-919.
26
BroekmanW, KhedoePPSJ, SchepersK, et al. Mesenchymal stromal cells: a novel therapy for the treatment of chronic obstructive pulmonary disease?[J]. Thorax, 2018, 73(6):565-574.
27
ChuangHM, ShihTE, LuKY, et al. Mesenchymal stem cell therapy of pulmonary fibrosis: improvement with target combination[J]. Cell Transplant, 2018, 27(11): 1581-1587.
28
Fernández ValloneVB, RomaniukMA, ChoiH, et al. Mesenchymal stem cells and their use in therapy: What has been achieved?[J]. Differentiation, 2013, 85(1-2):1-10.
29
Fayyad-KazanH, FaourWH, BadranB, et al. The immunomodulatory properties of human bone marrow-derived mesenchymal stromal cells are defined according to multiple immunobiological criteria[J]. Inflamm Res, 2016, 65(6):501-510.
30
ShiY, WangY, LiQ, et al. Immunoregulatory mechanisms of mesenchymal stem and stromal cells in inflammatory diseases[J]. Nat Rev Nephrol, 2018, 14(8):493-507.
31
WuZ, McGooganJM. Characteristics of and important lessons from the Coronavirus Disease 2019 (COVID-19) outbreak in China: summary of a report of 72314 cases from the Chinese center for disease control and prevention[J]. JAMA, 2020. doi: 10.1001/jama.2020.2648. Online ahead of print.
32
UccelliA, MorettaL, PistoiaV. Mesenchymal stem cells in health and disease[J]. Nat Rev Immunol, 2008,8(9):726-736.
33
NautaAJ, FibbeWE. Immunomodulatory properties of mesenchymal stromal cells[J]. Blood, 2007,110(10):3499-3506.
34
ZhouYY, YamamotoY, XiaoZD, et al. The immunomodulatory functions of mesenchymal stromal/stem cells mediated via paracrine activity[J]. J Clin Med, 2019, 8(7):1025.
35
GalipeauJ, SensébéL. Mesenchymal stromal cells: clinical challenges and therapeutic opportunities[J]. Cell Stem Cell, 2018, 22(6):824-833.
36
UccelliA, Nicole Kerlero deRosbo. The immunomodulatory function of mesenchymal stem cells: mode of action and pathways[J]. Ann N Y Acad Sci, 2015, 1351(1):114-126.
37
ZhengGP, GeMH, QiuG, et al. Mesenchymal stromal cells affect disease outcomes via macrophage polarization[J]. Stem Cells Int, 2015, 2015:989473.
38
ÐokićJM, TomićSZ, ČolićMJ. Cross-talk between mesenchymal stem/stromal cells and dendritic cells[J]. Curr Stem Cell Res Ther, 2016, 11(1): 51-65.
39
LiN, HuaJL. Interactions between mesenchymal stem cells and the immune system[J]. Cellular and Molecular Life Sciences, 2017, 74(13):2345-2360.
40
LiuGY, WangLH, PangTX, et al. Umbilical cord-derived mesenchymal stem cells regulate thymic epithelial cell development and function in Foxn1 (-/-) mice[J]. Cell Mol Immunol, 2014, 11(3):275-284.
41
LiB, ZhangH, ZengM, et al. Bone marrow mesenchymal stem cells protect alveolar macrophages from lipopolysaccharide-induced apoptosis partially by inhibiting the Wnt/β-catenin pathway[J].Cell Biol Int, 2015, 39(2):192-200.
42
LeeJW, FangX, GuptaN, et al. Allogeneic human mesenchymal stem cells for treatment of E. coli endotoxin-induced acute lung injury in the ex vivo perfused human lung[J]. Proc Natl Acad Sci U S A,2009, 106(38):16357-16362.
43
YiY, HuSL, XuXP, et al. The vascular endothelial growth factors-expressing character of mesenchymal stem cells plays a positive role in treatment of acute lung injury in vivo[J]. Mediators Inflamm, 2016, 2016: 2347938. Published online 2016 May 24. doi:10.1155/2016/2347938.
44
MeganO, MariusAM, ArulV, et al. Late rescue therapy with Cord-Derived mesenchymal stromal cells for established lung injury in experimental bronchopulmonary dysplasia[J]. Stem Cells Dev, 2020,29(6):364-371.
45
KwonJH, MiyeonK, YunKB, et al. Decorin secreted by human umbilical cord Blood-Derived mesenchymal stem cells induces macrophage polarization via CD44 to repair hyperoxic lung injury[J]. Int J Mol Sci, 2019, 20(19):4815.
46
HostettlerKE, GazdharA, KhanP, et al. Multipotent mesenchymal stem cells in lung fibrosis[J]. PLoS One, 2017, 12(8):e0181946.
47
El-TantawyHW, Ekram Nemr Abd AlHaleem. Therapeutic effects of stem cell on hyperglycemia, hyperlipidemia, and oxidative stress in alloxan-treated rats[J]. Mol Cell Biochem, 2014, 391(1/2):193-200.
48
RadwanSM, GhoneimD, SalemM, et al. Adipose tissue-derived mesenchymal stem cells protect against Amiodarone-Induced lung injury in rats[J]. Appl Biochem Biotechnol, 2020191(3):1027-1041.
49
MahmoudiT, KamalA, BashiriH, et al. Hydrogen peroxide preconditioning promotes protective effects of umbilical cord vein mesenchymal stem cells in experimental pulmonary fibrosis[J]. Adv Pharm Bull, 2020, 10(1):72-80.
50
RojasM, XuJ, WoodsCR, et al. Bone marrow-derived mesenchymal stem cells in repair of the injured lung[J]. Am J Respir Cell Mol Biol, 2005, 33(2):145-152.
51
NeuringerIP, RandellSH. Stem cells and repair of lung injuries[J]. Respir Res, 2004, 5(1):6.
52
ZhengG, HuangL, TongH, et al. Treatment of acute respiratory distress syndrome with allogeneic adipose-derived mesenchymal stem cells: a randomized, placebo-controlled pilot study[J]. Respir Res, 2014, 15(1):39.
53
WilsonJG, LiuKD, ZhuoH, et al. Mesenchymal stem (stromal) cells for treatment of ARDS:a phase 1 clinical trial[J]. Lancet Respir Med, 2015, 3(1):24-32.
54
MichaelAM, CarolynSC, ZhuoHJ, et al. Treatment with allogeneic mesenchymal stromal cells for moderate to severe acute respiratory distress syndrome (START study): a randomised phase 2a safety trial[J]. Lancet Respir Med, 2019, 7(2):154-162.
55
ChenJ, HuCX, ChenLJ, et al. Clinical study of mesenchymal stem cell treatment for acute respiratory distress syndrome induced by epidemic influenza a (H7N9) infection: a hint for COVID-19 treatment[J]. Engineering(Beijing), 2020. doi: 10.1016/j.eng.2020.02.006. Online ahead of print.
56
LengZK, ZhuRJ, WeiH, et al. Transplantation of ACE2- mesenchymal stem cells improves the outcome of patients with COVID-19 pneumonia[J]. Aging Dis, 2020, 11(2):216-228.
57
LiangB, ChenJH, LiT, et al. (2020)Clinical remission of a critically ill COVID-19 patient treated by human umbilical cord mesenchymal stem cells[J]. 2020, 9(2). doi:10.12074/202002.00084.
58
LiGD, ErikDC. Therapeutic options for the 2019 novel coronavirus (2019-nCoV)[J]. Nat Rev Drug Discov, 2020, 19(3):149-150.
59
中国细胞生物学学会干细胞生物学分会&中华医学会感染病学分会.《干细胞治疗新型冠状病毒肺炎(COVID-19)临床研究与应用专家指导意见》[EB/OL].(2020-03-27)[2020-06-23].http://www.most.gov.cn/gnwkjdt/202003/t20200327_152617.htm




