


Keynote Speech
Rina-S-the missing link(er)
Protein homeostasis by dual-precision targeted protein degradation and stabilization
A novel dual-payload ADC platform to overcome payload resistance and maximize therapeutic promise of ADCs
MYTX-011: a cMET-targeting ADC engineered for anti-tumor activity against a broader spectrum of cMET expression
NTX1105: development of a best-in-class ADC targeting Nectin-4
Collaborating within the global life science ecosystem to advance breakthrough science
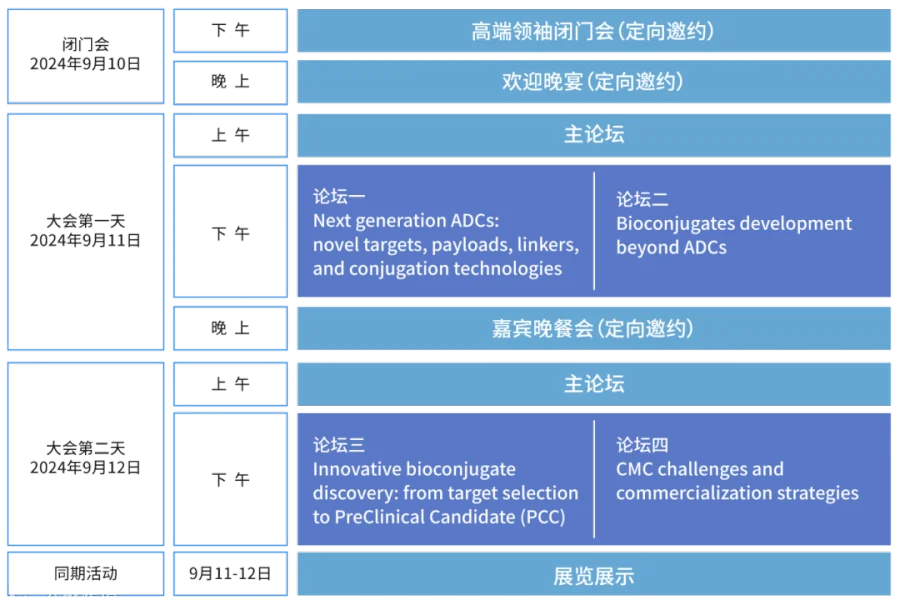
论坛一:Next generation ADCs: Novel targets, payloads, payload-linkers, and conjugation technologies
Next generation ADCs: novel targets, payloads, linkers and conjugation technologies
Nexatecan™: OHPAS-able Topo1 inhibitor for ADC
Introduction to PINOT-ADC platform: novel Top1 inhibitor payload, tandem cleavable and super-hydrophilic linker, and dual payload system
Versatile drug bundle-based ADC platform: achieving site-specific conjugation, DAR of 8 or 12, and dual payloads
CS5001, a potential best-in-class ROR1 ADC
Discovery of AT65474, a highly selective anti-CLDN6 ADC with a proprietary payload
Polysorbates in biopharmaceuticals-approaches to mitigate risk
论坛二:Bioconjugates development beyond ADCs
RDC-the exploration of new drug modality in unmet clinical need
Intra-cellular mutant epitopes-novel targets for ADC and bispecific antibodies?
Developing radiopharmaceuticals targeting CLDN 18.2 with nanobodies
Fully human common light chain technology for novel ADCs
论坛三:Innovative bioconjugate discovery: from target selection to PreClinical Candidate (PCC)
Harnessing AbClick Pro® for AT-211: leading CLDN 18.2 ADCs with superior therapeutic index
An overview on the new topoisomerases inhibitors technologies
Next generation ADCs:novel targets, payloads, linkers and conjugation technologies
Challenges and solutions for clinical PK bioanalysis of antibody drug conjugates (ADCs)
论坛四:CMC challenges and commercialization strategies
CMC scale-up challenges and COGs for site specific ADCs
Innovative bioassays: translate clinical effects into a rigorous system of XDC product assessment
The integrated analytical platform enables accelerated CMC development of XDCs
QbD considerations for ADC process
How WuXiBio’s Microbial Platform facilitates rapid and cost-effective ADC development and manufacturing
Non-clinical strategies for ADC drugs
更多确认嘉宾及议题持续公布中.....

主办方
药明合联WuXi XDC、佰傲谷BioValley
大会时间
2024年9月10日-12日
大会地点
江苏省无锡君来洲际酒店
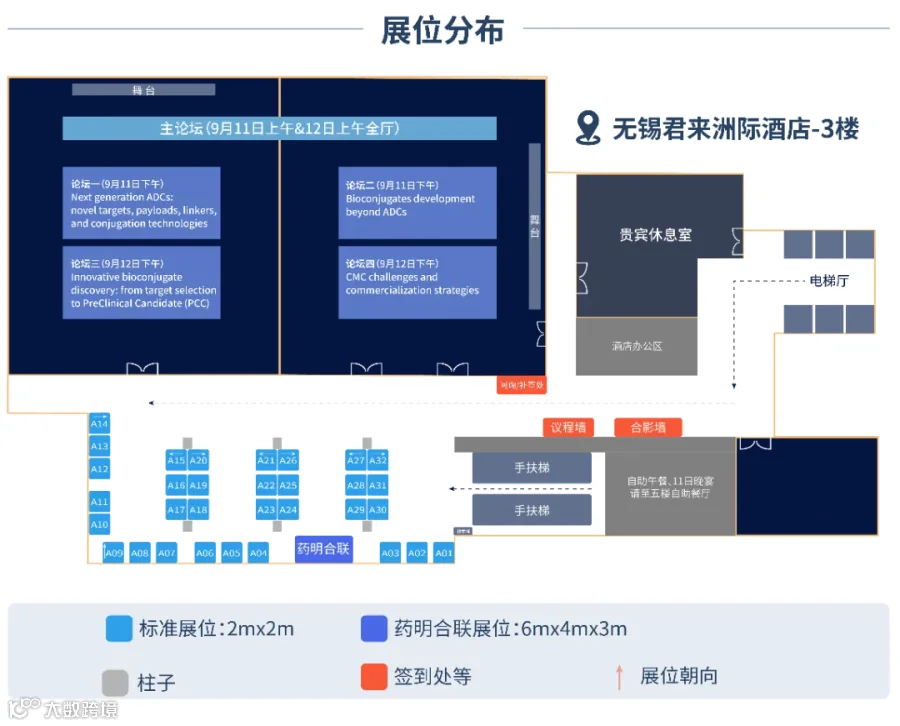
展位分布
报名方式

欢迎更多XDC研发企业加入(标准通票:200元/人)
温馨提示:如您收到主办方特别邀请,请通过特别邀请链接或者二维码进行注册登记,无需购买标准通票。
Global XDC 2024
诚邀拨冗出席
共拓行业未来


关于药明合联
药明合联生物技术有限公司(股票代码:2268.HK,以下简称"药明合联")是全球领先的生物偶联药合同研究、开发和生产企业(CRDMO),专注于提供抗体偶联药物(ADC)等生物偶联药端到端服务,涵盖抗体或其他偶联药中间体、连接子/化学有效载荷、偶联原液及制剂等研发和GMP生产领域。如需了解更多信息,请访问:www.wuxixdc.com

药明合联一站式平台
即刻扫码 关注我们


