
Swipe Left For English News
1
药明生物利用WuXiUPTM首次在中试规模实现了从灌流细胞培养到最终超滤/渗滤(UF/DF)的端到端连续原液生产
2
该端到端连续生产工艺也将推广至药明生物全球生产基地,并进一步应用于GMP生产环节
上海,
2023年11月22日
全球领先的合同研究、开发和生产(CRDMO)服务公司药明生物(WuXi Biologics, 2269.HK)宣布其在上海非GMP中试生产车间通过超高效连续生物工艺平台WuXiUPTM完成了中试规模的端到端原液生产。目前,该生物工艺正放大至GMP生产规模,未来也将在公司中国、爱尔兰、美国、新加坡等全球基地推广。
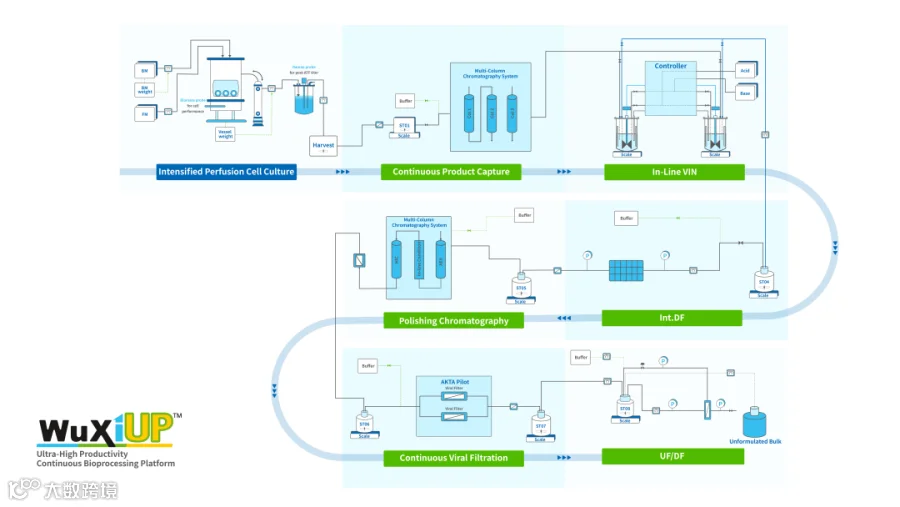
全连续工艺始于灌流细胞培养,收获液经历了完全连续的下游工艺,直至最终UF/DF环节
此次药明生物通过WuXiUPTM实现了重大突破:上游总时长25天的连续细胞培养总产量高达105克/升,其中在18天的抗体表达阶段,每日平均产量高达6克/升。在下游纯化工艺阶段,从Protein A捕获到UF/DF的总收率近80%,整个批次从40升培养体积中总计获得超过三千克蛋白。下游工艺开发使用了连续捕获,使得批次Protein A填料需求大大降低,显著降低临床生产阶段填料成本。同时,全连续的下游操作单元也降低了对厂房空间面积的要求。
为了确保实时监测工艺动态,加强工艺控制和在线产品放行,该连续生产过程中还应用了定制化的过程分析技术(PAT),以监测细胞浓度、表达量、纯度、浊度、单体纯度和蛋白浓度等指标。
通过WuXiUPTM端到端连续原液生产技术,应用1000升一次性生物反应器生产单抗可以实现每批次产量超过60公斤,每年产量达到约800公斤,与10000升至20000升传统生物反应器产能相当。这种行业领先的生物工艺实现了超高产量和卓越质量,将显著加快生物药上市进程,并且大幅降低商业化产品的生产成本。同时,由于该工艺对厂房空间面积要求降低,也为工艺操作提供了更高的灵活性。
陈智胜博士
首席执行官
药明生物

我们对于WuXiUPTM的突破感到非常自豪,这也是药明生物首次实现全连续原液生产。在全球范围内开发新一代生物药生产技术,提升生物药可及性始终是药明生物不懈奋斗的使命。随着WuXiUPTM全连续生产全面发力,我们也将继续践行加快和变革全球生物药开发和生产进程的使命,让更多患者用得到、用得起高质量生物药。

周伟昌博士
全球生物制药开发及运营总裁、首席技术官
药明生物

此次WuXiUPTM取得的成就彰显了药明生物不断深耕与精进以连续生产为代表的生物技术平台。通过这些领先的技术平台,结合引领行业的洞见和能力,我们正在以更高效的生物药开发和生产解决方案,满足全球客户多元化的服务需求。

关于WuXiUPTM
WuXiUPTM是药明生物针对加快生物药开发和生产进程,提高生物药可及性而自主研发的新一代生物药生产解决方案。
借助超高效连续生物工艺平台WuXiUPTM,包括单克隆抗体和融合蛋白在内的几乎所有生物药都能够实现超高产率,一款抗体Fc融合蛋白产品20天连续生产批次产量高达51克/升。该平台工艺开发速度与传统流加生产工艺相当,也可快速从传统流加工艺而来,并具有卓越的可放大性与稳健性。WuXiUPTM平台前瞻性地融合了连续细胞培养技术与连续柱层析技术,实现超越传统纯化工艺的高纯化收率。
推荐阅读
新闻速递 | 前沿洞见 | 精彩活动
· WuXiUP连续生产工艺平台大揭秘:日均产量超3g/L,27天高达86g/L!
· 批次产量高达51克/升!药明生物WuXiUP平台成功实现细胞培养重大突破




关于药明生物
药明生物(股票代码:2269.HK)是一家全球领先的合同研究、开发和生产(CRDMO)服务公司。公司通过开放式、一体化生物制药能力和技术赋能平台,提供全方位的端到端服务,帮助合作伙伴发现、开发及生产生物药,实现从概念到商业化生产的全过程,加速全球生物药研发进程,降低研发成本,造福病患。
药明生物在中国、美国、爱尔兰、德国和新加坡拥有超过12000名员工。通过药明生物人的专业服务团队,以及先进技术和精深洞见,公司为客户提供高效经济的生物药解决方案。截至2023年6月底,药明生物帮助客户研发和生产的综合项目高达621个,其中包括22个商业化生产项目。
药明生物将环境、社会和治理(ESG)视为业务发展和企业精神的重要组成部分,并致力于成为全球生物药CRDMO领域的ESG领导者,例如应用更绿色环保的新一代生物制药技术和能源等引领行业发展。公司成立了由首席执行官领导的ESG委员会,全面落实ESG战略并践行可持续性发展承诺。
更多信息,请访问:www.wuxibiologics.com


媒体关系 PR@wuxibiologics.com
业务垂询 info@wuxibiologics.com
WuXi Biologics Successfully Implemented a Fully Integrated Continuous Process with a Breakthrough Productivity of ~6 g/L/day at Pilot Scale
1
By leveraging WuXiUPTM, WuXi Biologics has completed its first end-to-end continuous drug substance (DS) manufacturing from perfused cell culture to final UF/DF pool at pilot scale
2
This end-to-end continuous bioprocess, which is in place for WuXi Biologics global manufacturing network, is currently being scaled up to GMP manufacturing
Shanghai,
November 22, 2023
WuXi Biologics ("WuXi Bio") (2269.HK), a leading global Contract Research, Development, and Manufacturing Organization (CRDMO), announced today that it has accomplished end-to-end DS manufacturing at pilot scale utilizing WuXiUPTM, a proprietary ultra-high productivity continuous bioprocessing platform at its non-GMP pilot plant in Shanghai, China. Currently, this well configured bioprocess is being scaled up to GMP manufacturing and will be deployed across WuXi Biologics' global manufacturing sites in China, Ireland, U.S., Singapore.
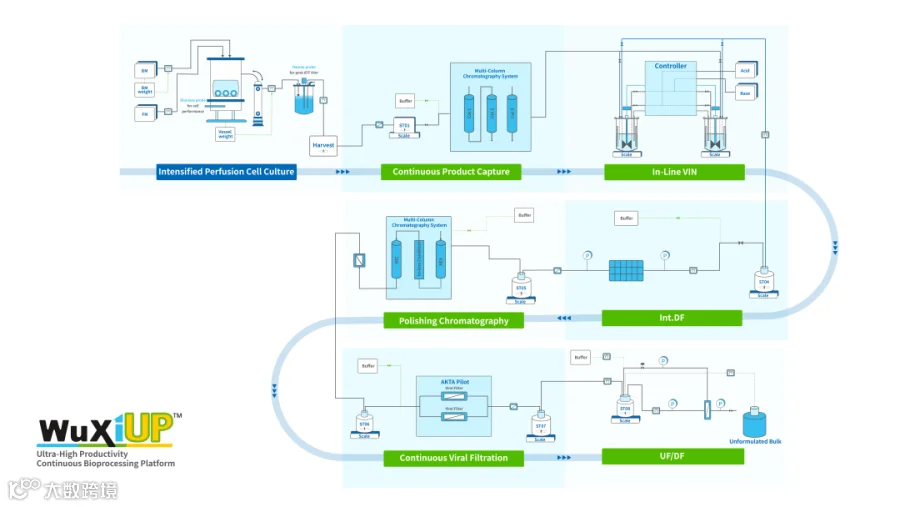
The continuous process started from perfused cell culture. The harvest fluid from upstream went through fully continuous downstream process to final UF/DF pool.
WuXi Biologics achieved a major breakthrough utilizing WuXiUPTM: An average daily productivity of 6 g/L/day for a mAb molecule was obtained during the 18-day production phase, leading to a significantly high accumulated volumetric productivity at 105 g/L in the 25-day continuous cell culture process. During the downstream process, the total yield calculated from Protein A capture to UF/DF reached nearly 80%, delivering over 3 kg protein from 40 L working volume. The continuous capture significantly reduced Protein A resin requirement per batch, which will significant lower the resin cost at clinical manufacturing stage. Meanwhile, the integrated downstream unit operations resulted in operational efficiency requiring minimal facility footprint utilization.
To ensure real-time process monitoring, facilitate process control and product release, this continuous manufacturing process also utilized custom-fit process analytical technology (PAT) to monitor cell concentration, titer, purity, turbidity, monomer purity, and protein concentration.
With the end-to-end continuous DS manufacturing enabled by WuXiUPTM, a 1,000 L disposable bioreactor can produce more than 60 kg of mAb per batch and approximately 800 kg per year, achieving productivity comparable to a traditional 10,000-20,000L bioreactor. This industry-leading bioprocessing platform achieves high productivity and premier product quality which will expedite biologics development and reduce the cost of goods (COGS) of commercial products while increasing operational flexibility due to the minimal requirements for facility footprint utilization.
Dr. Chris Chen
CEO
WuXi Biologics

We're proud to achieve fully integrated DS manufacturing utilizing WuXiUPTM for the first time. Developing next-generation manufacturing technologies to improve affordability of biologics is always aligned with our mission of accelerating and transforming how biologics are developed and manufactured within the global setting. With this breakthrough, we have advanced our capabilities to enable global partners to bring more affordable high-quality biologics to market for the benefits of patients worldwide.

Dr. Weichang Zhou
President of Global Biologics Development and Operations
CTO
WuXi Biologics

The achievement of WuXiUPTM reflects WuXi Biologics' consistent focus on enhancing technology platforms such as continuous bioprocessing to provide efficient and cost-effective solutions for global clients. With these leading technology platforms, our combined industry-leading expertise and capabilities allow us to facilitate more efficient biologics development and manufacturing to meet diverse clients' needs.

About WuXiUPTM
WuXiUPTM, a proprietary ultra-high productivity continuous bioprocessing platform, is designed by WuXi Biologics to accelerate biologics development and manufacturing as well as to improve biologics accessibility and affordability.
With this new continuous processing platform, ultra-high productivity can be achieved in almost all biologics including mAbs and fusion proteins, with cell culture time periods similar to batch processes. Using a propriety continuous cell culture, a productivity of 51g/L has been achieved, which is more than 10 times the productivity of a 3-5g/L traditional fed-batch process. Suitable for commercial-scale production, the breakthrough WuXiUPTM platform couples continuous cell culture operations with continuous column chromatography, achieving higher purification yields than traditional step-wise unit operation purification processes.




About WuXi Biologics
WuXi Biologics (stock code: 2269.HK) is a leading global Contract Research, Development and Manufacturing Organization (CRDMO) offering end-to-end solutions that enable partners to discover, develop and manufacture biologics – from concept to commercialization – for the benefit of patients worldwide.
With over 12,000 skilled employees in China, the United States, Ireland, Germany and Singapore, WuXi Biologics leverages its technologies and expertise to provide customers with efficient and cost-effective biologics discovery, development and manufacturing solutions. As of June 30, 2023, WuXi Biologics is supporting 621 integrated client projects, including 22 in commercial manufacturing.
WuXi Biologics views Environmental, Social, and Governance (ESG) responsibilities as an integral component of our ethos and business strategy, and we aim to become an ESG leader in the biologics CRDMO sector. Our facilities use next-generation biomanufacturing technologies and clean-energy sources. We have also established an ESG committee led by our CEO to steer the comprehensive ESG strategy and its implementation, enhancing our commitment to sustainability.
For more information about WuXi Biologics, please visit: www.wuxibiologics.com


Contacts
Business
info@wuxibiologics.com
Media
PR@wuxibiologics.com



注:本信息不构成药明生物的信息披露或投资建议



