click on the blue words above to pay attention
On 26th April 2018, EU member states had approved a European Commission proposal to restrict the use of carcinogenic, mutagenic and reprotoxic (CMR) substances in clothing, textiles and footwear. The proposal brings new limits to 33 CMRs.
The main contents are as follows:
1.Shall not be placed on the market after twenty-four months of the regulation into force in any of the following:
a)clothing or related accessories,
b)textiles other than clothing which, under normal or reasonably foreseeable conditionsof use, come into contact with human skin to an extent similar to clothing,
c)footwear, if the clothing, accessory, textile or footwear is for use by consumers and the substance is present in a concentration, measured in homogeneous material, greater than that specified for that substance in Appendix 12.
2.By way of derogation, in relation to the placing on the market of formaldehyde in jackets, coats or upholstery, the relevant concentration shall be 300 mg/kg during the period between twenty-four months and sixty months after the regulation into force. The concentration specified in Appendix 12 shall apply thereafter.
3.The Regulation includes exemptions. The restrictions will not apply to:
a)clothing, related accessories or footwear, or parts of clothing, related accessories or footwear, made exclusively of natural leather, fur or hide,
b)non-textile fasteners and non-textile decorative attachments,
c)second-hand clothing, related accessories, textiles other than clothing 3 or footwear,
d)clothing, related accessories, textiles or footwear within the scope of Regulation (EU) 2016/425 or (EU) 2017/745,
e)disposable textiles, 'disposable textiles' means textiles that are designed to be used only once or for a limited time and are not intended for subsequent use for the same or a similar purpose.
4.Restricted substances and maximum concentration limits by weight in homogeneous materials (Appendix12):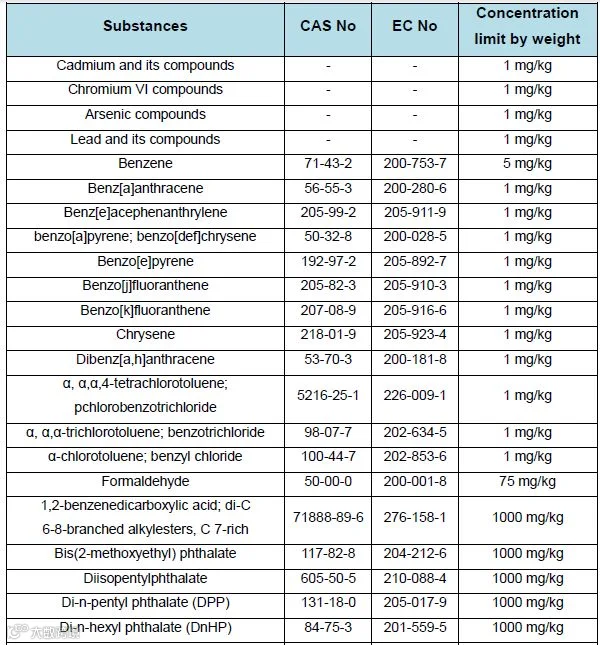
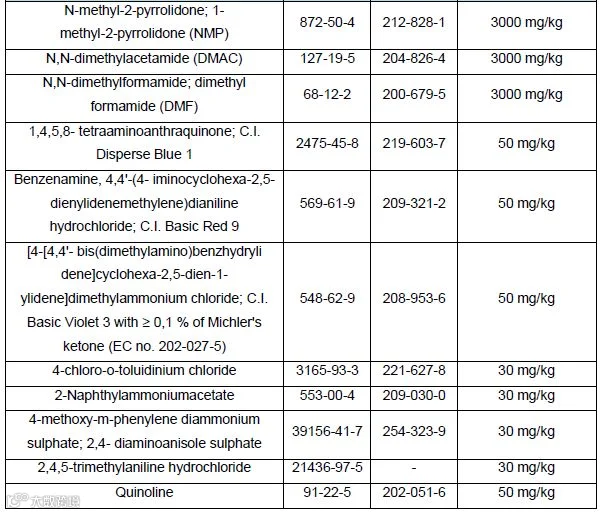
5.The new law will apply 24 months after publication in the EU Official Journal.
MTS reminds related companies to timely follow up on new laws and regulations, produce and export according to requirements, and pay attention to the requirements of the declaration of conformity. Scientific safety assessments must be carried out during the product design phase to the bulk production phase to ensure that relevant products meet the new regulatory requirements and avoid export risk

.




