click on the blue words above to pay attention

Oregon has adopted amendments to its children’s products reporting rule, which include modifications to its list of reportable substances and other changes to its fees and notification requirements.
The Toxic-Free Kids programme requires manufacturers of children’s products to notify the presence of substances included on the state’s High Priority Chemicals of Concern for Children’s Health (HPCCCH) list above de minimis levels.
The Oregon Health Authority (OHA) added five substances to the HPCCCH list, effective from 1 January, 2019. These are:
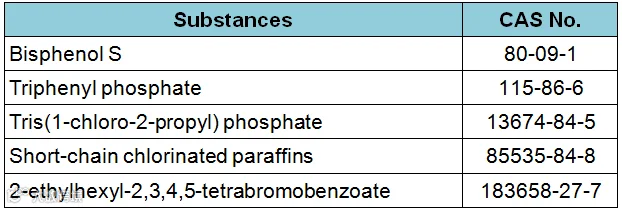
And it will remove the following three substances at that time:
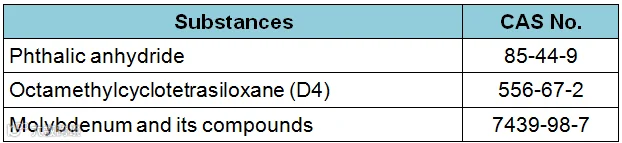
Oregon’s list of of reportable substances will total 68 when these changes come into force.
The OHA also adopted several modifications clarifying notification requirements and adjusting fee collection.
The updated rule says that notifications must include the number of children’s products offered for sale that contain a HPCCCH substance.
And it specifies that only one biennial report must be filed for a specific children’s product, and establishes a ‘hierarchy’ of entities which will be held responsible for this.
The product manufacturer sits at the top of this order of priority, unless that entity has no presence in the US. The distributor is next on the list, provided that it has a domestic presence. If it does not, then the responsibility falls to the importer.
These notification take effect from 1 October, 2018.

MTS reminds related companies to timely follow up on new laws and regulations, produce and export according to requirements, and pay attention to the requirements of the declaration of conformity. Scientific safety assessments must be carried out during the product design phase to the bulk production phase to ensure that relevant products meet the new regulatory requirements and avoid export risk.


长按二维码关注我们


