click on the blue words above to pay attention
20 April 2018, EU published a new RoHS stakeholder consultation to amend the RoHS Annex II list of restricted substances.
A stakeholder consultation has been launched as part of the “Study for the review of the list of restricted substances and to assess a new exemption request under Directive 2011/65/EU (RoHS 2.0) – Pack 15” and shall run for eight weeks, between 20 April 2018 to 15 June 2018.
The stakeholder consultation covers the collection of information for seven substances to be subjected to an assessment with a view to the review and amendment of the RoHS Annex II list of restricted substances.
The substances under review include:
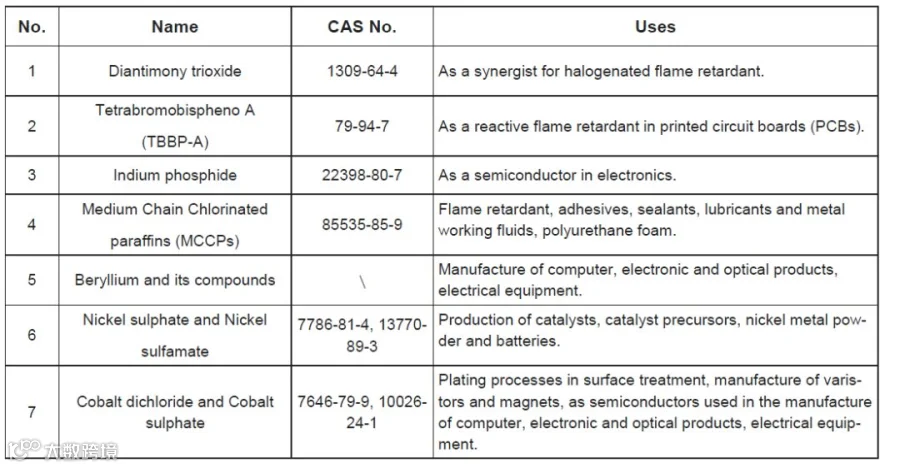

MTS reminds related companies to timely follow up on new laws and regulations, produce and export according to requirements, and pay attention to the requirements of the declaration of conformity. Scientific safety assessments must be carried out during the product design phase to the bulk production phase to ensure that relevant products meet the new regulatory requirements and avoid export risk.




