
ShiraYaacobi-Artzi,DoritKalo和ZviRoth*
以色列雷霍沃特希伯来大学农业、食品与环境罗伯特・H・史密斯学院动物科学系
摘要:胚胎移植已成为提高人类和牛生育能力的主要方法。本综述重点关注预测具有高发育能力和高妊娠潜力的胚胎。评估胚胎质量的一种方法是评估其形态,但这种方法提供的信息有限且不充分。使用延时系统可以对胚胎发育进行连续监测。本文介绍了主要的形态动力学参数,并讨论了它们预测胚胎活力、着床、妊娠和活产的潜力。特别关注了转录本谱与胚胎发育潜力之间的关联;正常卵裂的胚胎与其异常卵裂的对应胚胎在转录本谱上有所不同;同步和异步卵裂的胚胎(两者都被定义为适合移植)在转录本谱上也不同。最近,延时系统的进步和广泛使用导致了胚胎选择算法的发展。这项技术已经在人类体外受精单位中得到应用,但尚未用于家畜。作者认为,结合人类和牛的形态动力学信息可能揭示使用延时系统选择具有良好移植潜力的胚胎的好处。
关键词:形态动力学;牛胚胎;胚胎发育;延时系统;异常卵裂
1 引言
2 胚胎选择标准
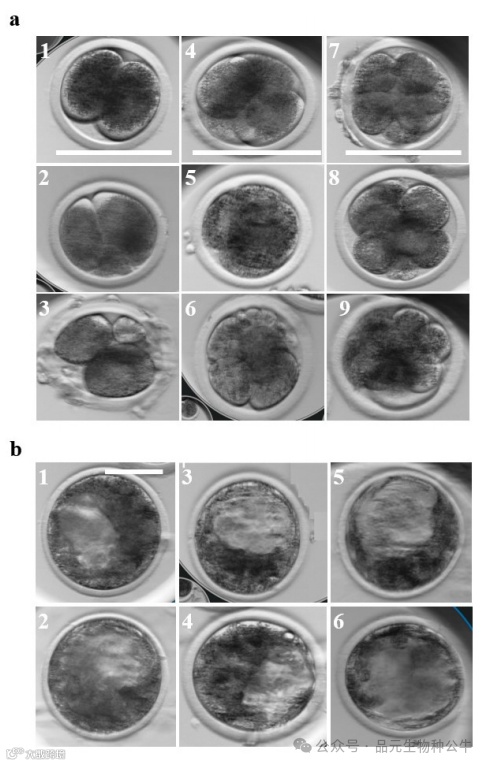
3 胚胎形态动力学

• 直接卵裂:一个卵裂球分裂成两个以上的子卵裂球[68,69]。
• 不等卵裂:产生大小不对称的卵裂球[14]。
• 反向卵裂:表现为卵裂球数量减少,很可能是由于卵裂球融合或胞质分裂失败[70]。

4 算法、机器学习和人工智能(AI)用于预测胚胎发育能力
5 总结
Kumaresan, A.; Srivastava, A.K. Frontier Technologies in Bovine Reproduction; Springer: Singapore, 2022.
Royal, M.D.; Darwash, A.O.; Flint, A.P.; Webb, R.; Woolliams, J.A.; Lamming, G.E. Declining fertility in dairy cattle: Changes in traditional and endocrine parameters of fertility. Anim. Sci. 2000, 70, 487–501.
Hansen, P.J.; Block, J. Towards an embryocentric world: The current and potential uses of embryo technologies in dairy production. Reprod. Fertil. Dev. 2003, 16, 1–14.
Wells, C.; Wiik, A.; Hanks, J.; Zavareh, A.; Killingsworth, R. Embryo morphokinetic activity evident in short videos of in vitro bovine embryos. Dairy 2022, 3, 849–861.
Osterman, M.J.; Hamilton, B.E.; Martin, J.A.; Driscoll, A.K.; Valenzuela, C.P. Births: Final data for 2020. Natl. Vital Stat. Rep. 2021, 70, 1–50.
Comolli, C.; Neyer, G.; Andersson, G.; Dommermuth, L.; Fallesen, P.; Jalovaara, M.; Jónsson, A.; Kolk, M.; Lappegard, T. Correction to: Beyond the Economic Gaze: Childbearing during and after Recessions in the Nordic Countries. Eur. J. Popul. 2021, 37, 473–520.
Centers for Disease Control and Prevention (CDC). 2021. Available online: https://www.cdc.gov/art/artdata/index.html (accessed on 13 June 2024).
Rabel, R.C.; Marchioretto, P.V.; Bangert, E.A.; Wilson, K.; Milner, D.J.; Wheeler, M.B. Pre-Implantation Bovine Embryo Evaluation—From Optics to Omics and Beyond. Animals 2023, 13, 2102.
Hansen, P.J. Some challenges and unrealized opportunities toward widespread use of the in vitro-produced embryo in cattle production. Animal 2023, 17, 100745.
Zhang, J.Q.; Li, X.L.; Peng, Y.; Guo, X.; Heng, B.C.; Tong, G.Q. Reduction in exposure of human embryos outside the incubator enhances embryo quality and blastulation rate. Reprod. BioMed Online 2010, 20, 510–515.
Gardner, D.K.; Meseguer, M.; Rubio, C.; Treff, N.R. Diagnosis of human preimplantation embryo viability. Hum. Reprod. Update 2015, 21, 727–747.
Arav, A.; Aroyo, A.; Yavin, S.; Roth, Z. Prediction of embryonic developmental competence by time-lapse observation and ‘shortest-half’ analysis. Reprod. Biomed. Online 2008, 17, 669–675.
Sugimura, S.; Akai, T.; Somfai, T.; Hirayama, M.; Aikawa, Y.; Ohtake, M.; Hattori, H.; Kobayashi, S.; Hashiyada, Y.; Konishi, K.; et al. Time-lapse cinematography-compatible polystyrene-based microwell culture system: A novel tool for tracking the development of individual bovine embryos. Biol. Reprod. 2010, 83, 97–978.
Sugimura, S.; Akai, T.; Hashiyada, Y.; Somfai, T.; Inaba, Y.; Hirayama, M.; Yamanouchi, T.; Matsuda, H.; Kobayashi, S.; Aikawa, Y.; et al. Promising system for selecting healthy in vitro-fertilized embryos in cattle. PLoS ONE 2012, 7, e36627.
Meseguer, M.; Herrero, J.; Tejera, A.; Hilligsøe, K.M.; Ramsing, N.B.; Remoh, J. The use of morphokinetics as a predictor of embryo implantation. Hum. Reprod. 2011, 26, 2658–2671.
Yaacobi-Artzi, S.; Kalo, D.; Roth, Z. Association between the morphokinetics of in-vitro-derived bovine embryos and the transcriptomic profile of the derived blastocysts. PLoS ONE 2022, 17, e0276642.
Giménez, C.; Conversa, L.; Murria, L.; Meseguer, M. Time-Lapse Imaging. Morphokinetic analysis of In Vitro Fertilization outcomes. Fertil. Steril. 2023, 120, 218–227.
Magata, F. Time-lapse monitoring technologies for the selection of bovine in vitro fertilized embryos with high implantation potential. Reprod. Dev. 2023, 69, 57–64.
Ozgur, K.; Berkkanoglu, M.; Bulut, H.; Donmez, L.; Isikli, A.; Coetzee, K. Blastocyst age, expansion, trophectoderm morphology, and number cryopreserved are variables predicting clinical implantation in single blastocyst frozen embryo transfers in freeze-only-IVF. J. Assist. Reprod. Genet. 2021, 38, 1077–1087.
Bakkensen, J.B.; Brady, P.; Carusi, D.; Romanski, P.; Thomas, A.M.; Racowsky, C. Association between blastocyst morphology and pregnancy and perinatal outcomes following fresh and cryopreserved embryo transfer. J. Assist. Reprod. Genet. 2019, 36, 2315–2324.
Ai, J.; Jin, L.; Zheng, Y.; Yang, P.; Huang, B.; Dong, X. The morphology of inner cell mass is the strongest predictor of live birth after a frozen-thawed single embryo transfer. Front. Endocrinol. 2021, 12, 621221.
Wang, X.; Zhang, S.; Gu, Y.; Ma, S.; Peng, Y.; Gong, F.; Tan, H.; Lin, G. The impact of blastocyst freezing and biopsy on the association of blastocyst morphological parameters with live birth and singleton birthweight. Fertil. Steril. 2023, 119, 56–66.
Montag, M.; van der Ven, H. Evaluation of pronuclear morphology as the only selection criterion for further embryo culture and transfer: Results of a prospective multicentre study. Hum. Reprod. 2001, 16, 2384–2389.
Scott, L. Pronuclear scoring as a predictor of embryo development. Reprod. Biomed. Online 2003, 6, 201–214.
Hardarson, T.; Hanson, C.; Sjögren, A.; Lundin, K. Human embryos with unevenly sized blastomeres have lower pregnancy and implantation rates: Indications for aneuploidy and multinucleation. Hum. Reprod. 2001, 16, 313–318.
Van Royen, E.V.; Mangelschots, K.; Vercruyssen, M.; Neubourg, D.D.; Valkenburg, M.; Ryckaert, G.; Gerris, J. Multinucleation in cleavage stage embryos. Hum. Reprod. 2003, 18, 1062–1069.
Gardner, D.K. Blastocyst culture: Toward single embryo transfers. Hum. Fertil. 2000, 3, 229–237.
Stringfellow, D.A.; Seide, S.M. Manual of the International Embryo Transfer Society: A Procedural Guide and General Information for the Use of Embryo Transfer Technology Emphasizing Sanitary Procedures, 3rd ed.; International Embryo Transfer Society: Savory, IL, USA, 1998.
Gardner, D.K.; Sakkas, D. Assessment of embryo viability: The ability to select a single embryo for transfer—A review. Placenta 2003, 24, S5–S12.
Farin, P.W.; Slenning, B.D.; Britt, J.H. Estimates of pregnancy outcomes based on selection of bovine embryos produced in vivo or in vitro. Theriogenology 1999, 52, 659–670.
Guerif, F.; Le Gouge, A.; Giraudeau, B.; Poindron, J.; Bidault, R.; Gasnier, O.; Royere, D. Limited value of morphological assessment at days 1 and 2 to predict blastocyst development potential: A prospective study based on 4042 embryos. Hum. Reprod. 2007, 22, 1973–1981.
Lemmen, J.G.; Agerholm, I.; Ziebe, S. Kinetic markers of human embryo quality using time-lapse recordings of IVF/ICSI-fertilized oocytes. Reprod. Biomed. Online 2008, 17, 385–391.
Wong, C.C.; Loewke, K.E.; Bossert, N.L.; Behr, B.; De Jonge, C.J.; Baer, T.M.; Pera, R.A. Non-invasive imaging of human embryos before embryonic genome activation predicts development to the blastocyst stage. Nat. Biotechnol. 2010, 28, 1115–1121.
Mizobe, Y.; Akiyoshi, T.; Minami, S.; Matsuo, K.; Fukushima, R.; Yamaguchi, A.; Okamoto, S. Effect of a time-lapse incubator (EmbryoScope®) on in vitro culture of human embryos. J. Mamm. Ova Res. 2014, 31, 40–44.
Goodman, L.R.; Goldberg, J.; Falcone, T.; Austin, C.; Desai, N. Does the addition of time-lapse morphokinetics in the selection of embryos for transfer improve pregnancy rates? A randomized controlled trial. Fertil. Steril. 2016, 105, 275–285.
Motato, Y.; de los Santos, M.J.; Escriba, M.J.; Ruiz, B.A.; Remohí, J.; Meseguer, M. Morphokinetic analysis and embryonic prediction for blastocyst formation through an integrated time-lapse system. Fertil. Steril. 2016, 105, 376–384.
Huang, T.T.F.; Chinn, K.; Kosasa, T.; Ahn, H.J.; Kessel, B. Morphokinetics of human blastocyst expansion in vitro. Reprod. Biomed. Online 2016, 33, 659–667.
Payne, D.; Flaherty, S.P.; Barry, M.F.; Matthews, C.D. Preliminary observations on polar body extrusion and pronuclear formation in human oocytes using time-lapse video cinematography. Hum. Reprod. 1997, 12, 532–541.
Shoukir, Y.; Campana, A.; Farley, T.; Sakkas, D. Early cleavage of in-vitro fertilized human embryos to the 2-cell stage: A novel indicator of embryo quality and viability. Hum. Reprod. 1997, 12, 1531–1536.
Van Soom, A.; Ysebaert, M.T.; de Kruif, A. Relationship between timing of development, morula morphology, and cell allocation to inner cell mass and trophectoderm in in vitro-produced bovine embryos. Mol. Reprod. Dev. 1997, 47, 47–56.
Dinnyés, A.; Lonergan, P.; Fair, T.; Boland, M.P. Timing of the first cleavage post-insemination affects cryosurvival of in vitro-produced bovine blastocysts. Mol. Reprod. Dev. 1999, 53, 318–324.
Lundin, K.; Bergh, C.; Hardarson, T. Early embryo cleavage is a strong indicator of embryo quality in human IVF. Hum. Reprod. 2001, 16, 2652–2657.
Fenwick, J.; Platteau, P.; Murdoch, A.P.; Herbert, M. Time from insemination to first cleavage predicts developmental competence of human preimplantation embryos in vitro. Hum. Reprod. 2002, 17, 407–412.
Somfai, T.; Inaba, Y.; Aikawa, Y.; Ohtake, M.; Kobayashi, S.; Konishi, K.; Imai, K. Relationship between the length of cell cycles, cleavage pattern and developmental competence in bovine embryos generated by in vitro fertilization or parthenogenesis. J. Reprod. Dev. 2010, 56, 200–207.
Gendelman, M.; Aroyo, A.; Yavin, S.; Roth, Z. Seasonal effects on gene expression, cleavage timing, and developmental competence of bovine preimplantation embryos. Reproduction 2010, 140, 73–82.
Lonergan, P.; Kahtir, H.; Piumi, F.; Rieger, D.; Humblot, P.; Boland, M.P. Effect of time interval from insemination to first cleavage on the developmental characteristics, sex ratio and pregnancy rate after transfer of bovine embryos. Reproduction 1999, 117, 159–167.
Holm, P.; Shukri, N.N.; Vajta, G.; Booth, P.; Bendixen, C.; Callesen, H. Developmental kinetics of the first cell cycles of bovine in vitro produced embryos in relation to their in vitro viability and sex. Theriogenology 1998, 50, 1285–1299.
Tsai, Y.C.; Chung, M.T.; Sung, Y.H.; Tsai, T.F.; Tsai, Y.T.; Lin, L.Y. Clinical value of early cleavage embryo. Int. J. Gynecol. Obstet. 2002, 76, 293–297.
Salumets, A.; Hydén-Granskog, C.; MaÈkinen, S.; Suikkari, A.M.; Tiitinen, A.; Tuuri, T. Early cleavage predicts the viability of human embryos in elective single embryo transfer procedures. Hum. Reprod. 2003, 18, 821–825.
Van Montfoort, A.P.; Dumoulin, J.C.; Kester, A.D.; Evers, J.L. Early cleavage is a valuable addition to existing embryo selection parameters: A study using single embryo transfers. Hum. Reprod. 2004, 19, 2103–2108.
Çiray, H.N.; Karagenç, L.; Uluǧ, U.; Bener, F.; Bahçeci, M. Early cleavage morphology affects the quality and implantation potential of day 3 embryos. Fertil. Steril. 2006, 85, 358–365.
Sakkas, D.; Shoukir, Y.; Chardonnens, D.; Bianchi, P.G.; Campana, A. Early cleavage of human embryos to the two-cell stage after intracytoplasmic sperm injection as an indicator of embryo viability. Hum. Reprod. 1998, 13, 182–187.
Sakkas, D.; Percival, G.; D’Arcy, Y.; Sharif, K.; Afnan, M. Assessment of early cleaving in vitro fertilized human embryos at the 2-cell stage before transfer improves embryo selection. Fertil. Steril. 2001, 76, 1150–1156.
Salumets, A.; Hydén-Granskog, C.; Suikkari, A.M.; Tiitinen, A.; Tuuri, T. The predictive value of pronuclear morphology of zygotes in the assessment of human embryo quality. Hum. Reprod. 2001, 16, 2177–2181.
Milazzotto, M.P.; Goissis, M.D.; Chitwood, J.L.; Annes, K.; Soares, C.A.; Ispada, J.; Assumpção, M.E.; Ross, P.J. Early cleavages influence the molecular and the metabolic pattern of individually cultured bovine blastocysts. Mol. Reprod. Dev. 2016, 83, 324–336.
Silva, T.; Santos, E.C.; Annes, K.; Soares, C.A.; Leite, R.F.; Lima, C.B.; Milazzotto, M.P. Morphokinetic-related response to stress in individually cultured bovine embryos. Theriogenology 2016, 86, 1308–1317.
El-Sayed, A.; Hoelker, M.; Rings, F.; Salilew, D.; Jennen, D.; Tholen, E.; Sirard, M.A.; Schellander, K.; Tesfaye, D. Large-scale transcriptional analysis of bovine embryo biopsies in relation to pregnancy success after transfer to recipients. Physiol. Genom. 2006, 28, 84–96.
Gutiérrez-ad’n, A.; Rizos, D.; Fair, T.; Moreira, P.N.; Pintado, B.; Fuente, J.D.; Boland, M.P.; Lonergan, P. Effect of speed of development on mRNA expression pattern in early bovine embryos cultured in vivo or in vitro. Mol. Reprod. Dev. 2004, 68, 441–448.
Liu, Z.; Xiang, G.; Xu, K.; Che, J.; Xu, C.; Li, K.; Wang, B.; Mu, Y. Transcriptome analyses reveal differential transcriptional profiles in early-and late-dividing porcine somatic cell nuclear transfer embryos. Genes 2020, 11, 1499.
Brevini, T.A.L.; Lonergan, P.; Cillo, F.; Francisci, C.; Favetta, L.A.; Fair, T.; Gandolfi, F. Evolution of mRNA polyadenylation between oocyte maturation and first embryonic cleavage in cattle and its relation with developmental competence. Mol. Reprod. Dev. 2002, 63, 510–517.
Orozco-Lucero, E.; Dufort, I.; Robert, C.; Sirard, M.A. Rapidly cleaving bovine two-cell embryos have better developmental potential and a distinctive mRNA pattern. Mol. Reprod. Dev. 2014, 81, 31–41.
Song, X.; Li, T.; Xiong, X.; Shan, H.; Feng, T.; Cui, K.; Shi, D.; Liu, Q.; Li, Z. RNA-Seq Reveals the underlying molecular mechanism of first cleavage time affecting porcine embryo development. Genes 2022, 13, 1251.
Ispada, J.; De Lima, C.B.; Sirard, M.A.; Fontes, P.K.; Nogueira, M.F.G.; Annes, K.; Milazzotto, M.P. Genome-wide screening of DNA methylation in bovine blastocysts with different kinetics of development. Epigenetics Chromatin 2018, 11, 1.
Basile, N.; Vime, P.; Florensa, M.; Aparicio Ruiz, B.; Garcia Velasco, J.A.; Remohí, J.; Meseguer, M. The use of morphokinetics as a predictor of implantation: A multicentric study to define and validate an algorithm for embryo selection. Hum. Reprod. 2015, 30, 276–283.
Liu, Y.; Chapple, V.; Feenan, K.; Roberts, P.; Matson, P. Time-lapse deselection model for human day 3 in vitro fertilization embryos: The combination of qualitative and quantitative measures of embryo growth. Fertil. Steril. 2016, 105, 656–662.
Grisart, B.; Massip, A.; Dessy, F. Cinematographic analysis of bovine embryo development in serum-free oviduct-conditioned medium. Reproduction 1994, 34, 399–405.
Holm, P.; Booth, P.J.; Callesen, H. Kinetics of early in vitro development of bovine in vivo-and in vitro-derived zygotes produced and/or cultured in chemically defined or serum-containing media. Reproduction 2002, 123, 553–565.
Zhan, Q.; Ye, Z.; Clarke, R.; Rosenwaks, Z.; Zaninovic, N. Direct unequal cleavages: Embryo developmental competence, genetic constitution and clinical outcome. PLoS ONE 2016, 11, e0166398.
Nagai, H.; Okada, M.; Nagai, Y.; Sakuraba, Y.; Okae, H.; Suzuki, R.; Sugimura, S. Abnormal cleavage is involved in the self-correction of bovine preimplantation embryos. Biochem. Biophys. Res. Commun. 2021, 562, 76–82.
Athayde Wirka, K.; Chen, A.A.; Conaghan, J.; Ivani, K.; Gvakharia, M.; Behr, B.; Suraj, V.; Tan, L.; Shen, S. Atypical embryo phenotypes identified by time-lapse microscopy: High prevalence and association with embryo development. Fertil. Steril. 2014, 101, 1637–1648.
Lechniak, D.; Sell-Kubiak, E.; Warzych, E. The metabolic profile of bovine blastocysts is affected by in vitro culture system and the pattern of first zygotic cleavage. Theriogenology 2022, 188, 43–51.
Liu, Y.; Chapple, V.; Roberts, P.; Matson, P. Prevalence, consequence, and significance of reverse cleavage by human embryos viewed with the use of the Embryoscope time-lapse video system. Fertil. Steril. 2014, 102, 1295–1300.e2.
Yang, S.T.; Shi, J.X.; Gong, F.; Zhang, S.P.; Lu, C.F.; Tan, K.; Leng, L.Z.; Hao, M.; He, H.; Gu, Y.F.; et al. Cleavage pattern predicts developmental potential of day 3 human embryos produced by IVF. Reprod. Biomed. Online 2015, 30, 625–634.
Rubio, I.; Kuhlmann, R.; Agerholm, I.; Kirk, J.; Herrero, J.; Escribá, M.J.; Bellver, J.; Meseguer, M. Limited implantation success of direct-cleaved human zygotes: A time-lapse study. Fertil. Steril. 2012, 98, 1458–1463.
Barrie, A.; Homburg, R.; McDowell, G.; Brown, J.; Kingsland, C.; Troup, S. Preliminary investigation of the prevalence and implantation potential of abnormal embryonic phenotypes assessed using time-lapse imaging. Reprod. Biomed. Online 2017, 34, 455–462.
Li, L.; Zheng, P.; Dean, J. Maternal control of early mouse development. Development 2010, 137, 859–870.
Yakovenko, S.A.; Apryshko, V.P.; Seregina, E.A.; Yutkin, E.V. Blastomere cleavage synchronicity evaluation as a tool in non-invasive selection of euploid embryos with high development competence. Fertil. Steril. 2011, 96, S86.
Wiener-Megnazi, Z.; Fridman, M.; Koifman, M.; Lahav-Baratz, S.; Stein, N.; Auslender, R.; Dirnfeld, M. Synchronous and asynchronous blastomere cleavage at cryopreservation: Effect on subsequent embryo survival, pregnancy and live birth rates. J. Biomed. Sci. Eng. 2014, 7, 243.
Balaban, B.; Brison, D.; Calderon, G.; Catt, J.; Conaghan, J.; Cowan, L.; Ebner, T.; Gardner, D.; Hardarson, T.; Lundin, K.; et al. Istanbul consensus workshop on embryo assessment: Proceedings of an expert meeting. Reprod. Biomed. Online 2011, 22, 632–646.
Magata, F.; Ideta, A.; Okubo, H.; Matsuda, F.; Urakawa, M.; Oono, Y. Growth potential of bovine embryos presenting abnormal cleavage observed through time lapse cinematography. Theriogenology 2019, 133, 119–124.
McCollin, A.; Swann, R.L.; Summers, M.C.; Handyside, A.H.; Ottolini, C.S. Abnormal cleavage and developmental arrest of human preimplantation embryos in vitro. Eur. J. Med. Genet. 2020, 63, 103651.
Lagalla, C.; Tarozzi, N.; Sciajno, R.; Wells, D.; Di Santo, M.; Nadalini, M.; Distratis, V.; Borini, A. Embryos with morphokinetic abnormalities may develop into euploid blastocysts. Reprod. Biomed. Online 2017, 34, 137–146.
Shavit, M.; Gonen, D.; Atzmon, Y.; Aslih, N.; Bilgory, A.; Shibli Abu-Raya, Y.; Sharqawi, M.; Estrada Garcia, D.; Michaeli, M.; Polotov, D.; et al. Cleavage patterns of 9600 embryos: The importance of irregular cleavage. J. Clin. Med. 2023, 12, 5656.
Daughtry, B.L.; Rosenkrantz, J.L.; Lazar, N.H.; Fei, S.S.; Redmayne, N.; Torkenczy, K.A.; Adey, A.; Yan, M.; Gao, L.; Park, B.; et al. Single-cell sequencing of primate preimplantation embryos reveals chromosome elimination via cellular fragmentation and blastomere exclusion. Genome Res. 2019, 29, 367–382.
Scott, L.; Finn, A.; O’leary, T.; McLellan, S.; Hill, J. Morphologic parameters of early cleavage-stage embryos that correlate with fetal development and delivery: Prospective and applied data for increased pregnancy rates. Hum. Reprod. 2007, 22, 230–240.
Madeja, Z.E.; Pawlak, P.; Piliszek, A. Beyond the mouse: Non-rodent animal models for study of early mammalian development and biomedical research. Int. J. Dev. Biol. 2019, 63, 187–201.
Zhu, F.; Lu, X.; Jiang, Y.; Wang, D.; Pan, L.; Jia, C.; Zhang, L.; Xie, Y.; Zhao, M.; Liu, H.; et al. Proteomics reveals the underlying mechanism by which the first uneven division affects embryonic development in pig. Theriogenology 2023, 210, 42–52.
Tesarik, J.; Kopecny, V.; Plachot, M.; Mandelbaum, J. Ultrastructural and autoradiographic observations on multinucleated blastomeres of human cleaving embryos obtained by in-vitro fertilization. Hum. Reprod. 1987, 2, 127–136.
Winston, N.J.; Johnson, M.H.; Braude, P.R. Assessment of the cellular DNA content of whole mounted mouse and human oocytes and of blastomeres containing single or multiple nuclei. Zygote 1993, 1, 17–25.
Tao, R.; Bi, J.; Zhu, F.; Wang, X.; Jia, C.; Xu, H.; He, X.; Li, J. Division behaviors and their effects on pre-implantation development of pig embryos. Reprod. Domest. Anim. 2022, 57, 1016–1028.
Yang, T.; Yuan, X.; Xue, Q.; Sun, L.; Xu, T.; Chen, Y.; Shi, D.; Li, X. Comparison of symmetrical and asymmetrical cleavage 2-cell embryos of porcine by Smart-seq2. Theriogenology 2023, 210, 221–226.
Desai, N.; Ploskonka, S.; Goodman, L.R.; Austin, C.; Goldberg, J.; Falcone, T. Analysis of embryo morphokinetics, multinucleation and cleavage anomalies using continuous time-lapse monitoring in blastocyst transfer cycles. Reprod. Biol. Endocrinol. 2014, 12, 54.
Jin, L.; Dong, X.; Tan, W.; Huang, B. Incidence, dynamics and recurrences of reverse cleavage in aneuploid, mosaic and euploid blastocysts, and its relationship with embryo quality. J. Ovarian Res. 2022, 15, 91.
Petersen, B.M.; Boel, M.; Montag, M.; Gardner, D.K. Development of a generally applicable morphokinetic algorithm capable of predicting the implantation potential of embryos transferred on Day 3. Hum. Reprod. 2016, 31, 2231–2244.
Yang, S.H.; Wu, C.H.; Chen, Y.C.; Yang, C.K.; Wu, T.H.; Chen, P.C.; Tsai, H.D. Effect of morphokinetics and morphological dynamics of cleavage stage on embryo developmental potential: A time-lapse study. Taiwan J. Obstet. Gynecol. 2018, 57, 76–82.
Huayhua, C.; Rodríguez, M.; Vega, J.; Briones, M.; Rodriguez-Alvarez, L.; Mellisho, E. Blastulation time measured with time-lapse system can predict in vitro viability of bovine blastocysts. PLoS ONE 2023, 18, e0289751.
Theilgaard Lassen, J.; Fly Kragh, M.; Rimestad, J.; Nygård Johansen, M.; Berntsen, J. Development and validation of deep learning-based embryo selection across multiple days of transfer. Sci. Rep. 2023, 13, 4235.
Gardner, D.K.; Sakkas, D. Making and selecting the best embryo in the laboratory. Fertil. Steril. 2023, 120, 457–466.
Ueno, S.; Berntsen, J.; Ito, M.; Uchiyama, K.; Okimura, T.; Yabuuchi, A.; Kato, K. Pregnancy prediction performance of an annotation-free embryo scoring system on the basis of deep learning after single vitrified-warmed blastocyst transfer: A single-center large cohort retrospective study. Fertil. Steril. 2021, 116, 1172–1180.
Glatstein, I.; Chavez-Badiola, A.; Curchoe, C.L. New frontiers in embryo selection. J. Assist. Reprod. Genet. 2023, 40, 223–234.
Kato, K.; Ueno, S.; Berntsen, J.; Kragh, M.F.; Okimura, T.; Kuroda, T. Does embryo categorization by existing artificial intelligence, morphokinetic or morphological embryo selection models correlate with blastocyst euploidy rates? Reprod. Biomed. Online 2023, 46, 274–281.
Chavez-Badiola, A.; Farías, A.F.; Mendizabal-Ruiz, G.; Silvestri, G.; Griffin, D.K.; Valencia-Murillo, R.; Drakeley, A.J.; Cohen, J. Use of artificial intelligence embryo selection based on static images to predict first trimester pregnancy loss. Reprod. Biomed. Online 2024, 49, 103934.
VerMilyea, M.; Hall, J.M.; Diakiw, S.M.; Johnston, A.; Nguyen, T.; Perugini, D.; Miller, A.; Picou, A.; Murphy, A.P.; Perugini, M. Development of an artificial intelligence-based assessment model for prediction of embryo viability using static images captured by optical light microscopy during IVF. Hum. Reprod. 2020, 35, 770–784.
Jiang, V.S.; Bormann, C.L. Noninvasive genetic screening: Current advances in artificial intelligence for embryo ploidy prediction. Fertil. Steril. 2023, 120, 228–234.
Rocha, J.C.; Passalia, F.J.; Matos, F.D.; Takahashi, M.B.; Maserati, M.P., Jr.; Alves, M.F.; De Almeida, T.G.; Cardoso, B.L.; Basso, A.C.; Nogueira, M.F. Data descriptor: Automatized image processing of bovine blastocysts produced in vitro for quantitative variable determination. Sci. Data 2017, 4, 170192.
De Souza Ciniciato, D.; Takahashi, M.B.; Nogueira, M.F.G.; Rocha, J.C. Potential use of smartphone as a tool to capture embryo digital images from stereomicroscope and to evaluate them by an artificial neural network. In Proceedings of the International Conference on Computer-Human Interaction Research and Applications (CHIRA 2017), Funchal, Portugal, 31 October 31–2 November 2017; pp. 185–189.
Turki, T.; Wei, Z. Improved deep convolutional neural networks via boosting for predicting the quality of in vitro bovine embryos. Electronics 2022, 11, 1363.



译者/张逸松
审核/朱凯
审核/李雪宾


